The End of Knee Replacements? Stanford’s New Anti-Aging Cartilage Injection
At a Glance
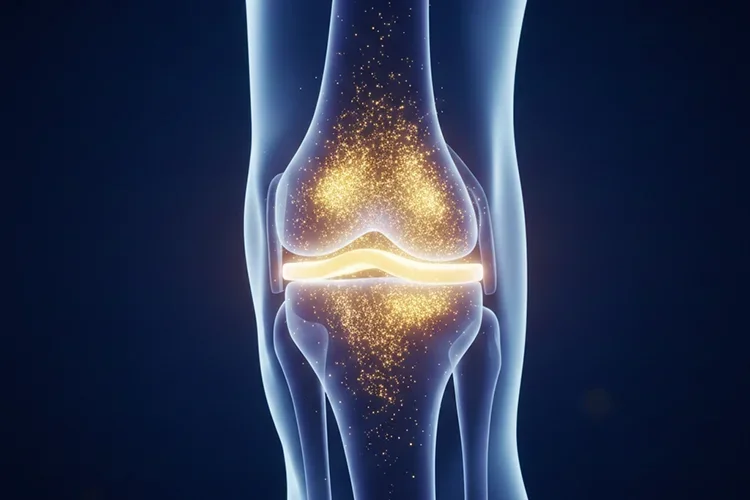
Click a point below to jump to the details.Regenerative medicine offers a non-invasive way to repair joints without surgery. (Image: AI-generated)
Our knees are masterpieces of nature, yet they are surprisingly fragile because cartilage doesn't have the power to heal on its own.
Once the smooth, shock-absorbing tissue known as hyaline cartilage is eroded by age or traumatic injury, the body often attempts to repair it with fibrocartilage, a stiff, inferior fibrous tissue.
This biological "patchwork" inevitably leads to osteoarthritis (OA), a debilitating condition that currently affects over 32.5 million adults in the U.S. alone.
For decades, the medical community has focused on managing symptoms—using anti-inflammatories or lubricant injections—while the patient essentially "waits" for the joint to fail enough to justify a total knee replacement.
The Science of "Gerozymes": Targeting 15-PGDH
At the heart of this discovery is a specific enzyme called 15-PGDH, which researchers have identified as a "gerozyme" (an enzyme associated with aging). Under normal youthful conditions, our bodies maintain healthy levels of Prostaglandin E2 (PGE2), a vital signaling molecule that promotes tissue repair and cellular health.
However, as we age or experience acute trauma, 15-PGDH levels spike significantly, leading to the rapid breakdown of PGE2. This creates a hostile environment where cartilage repair becomes impossible.
Cellular Rejuvenation
The treatment allows existing chondrocytes (cartilage cells) to revert to a youthful state, enabling them to synthesize new matrix.
Structural Integrity
Unlike previous attempts at regeneration, this method produces functional hyaline cartilage, which possesses the necessary elasticity and durability for high-impact movement.
Non-Invasive Delivery
The vision is a simple, targeted injection directly into the joint space, avoiding the risks and recovery times associated with open surgery.
The "Golden Hour" of Injury: Preventing Post-Traumatic Arthritis
Perhaps the most transformative aspect of this research is its potential in preventing post-traumatic osteoarthritis (PTOA). For young athletes, an ACL tear or meniscus injury is often a ticking clock; even with perfect surgical repair, the initial trauma triggers an inflammatory cascade that often leads to arthritis within 10 to 15 years.
Stanford’s researchers demonstrated that if the 15-PGDH inhibitor is administered shortly after a major injury, it can stabilize the joint’s biochemical environment. By preventing the initial degradation of cartilage, this injection acts as a biological shield, potentially "immunizing" the joint against the long-term risk of chronic disease. This could mean the difference between a 20-year-old athlete having a healthy knee at 50, or requiring a prosthetic joint before middle age.
Frequently Asked Questions
-
As of February 2026, while the results in animal models are groundbreaking, the treatment is moving toward human clinical trials. It typically takes several years to ensure safety and efficacy, but since similar 15-PGDH inhibitors have already passed Phase 1 safety trials for other conditions, the timeline may be accelerated compared to entirely new drugs.
-
The primary goal of this therapy is to regenerate existing cartilage cells. In severe Stage 4 cases where the cartilage is entirely gone, there may not be enough biological material left to stimulate. However, researchers are hopeful it can significantly delay or even eliminate the need for surgery in patients with mild to moderate degradation.
-
Currently, the research focuses on therapeutic and preventive restoration after a cellular aging signal (like 15-PGDH) has been triggered by injury or age. It is not intended as a "performance enhancer" for healthy joints. However, for athletes with minor wear-and-tear or early inflammatory signs, it may serve as a protective intervention to ensure their joints remain resilient throughout their career.
A Future Beyond the Scalpel
We are witnessing a shift from mechanical orthopedics to molecular orthopedics. The implications for longevity and quality of life are immense. If we can successfully transition this therapy to human clinical trials, the standard of care for joint health will be fundamentally altered.
Instead of accepting joint decay as an inevitable part of the human experience, we may soon treat it with the same precision we apply to other biological conditions.
Sources & References
- [1] SciTechDaily (2026). "Anti-Aging Injection Regrows Knee Cartilage and Prevents Arthritis." SciTechDaily Reports.
- [2] Singla, M., Wang, Y. X., et al. (2025). "Inhibition of 15-hydroxy prostaglandin dehydrogenase promotes cartilage regeneration." Science.
- [3] Palla, A. R., et al. (2021). "Inhibition of 15-PGDH rejuvenates muscle mass and function in aged mice." Science.
Disclaimer: This article explores emerging healthcare trends and experimental studies for educational insights. It is not a substitute for professional medical advice or a guarantee of clinical outcomes. This content is for informational purposes only. Some imagery is AI-generated for educational clarity. Copyright © 2026 TheWellnessExaminer. All rights reserved.